Acute compartment syndrome (ACS) is a true orthopedic emergency [1] that evolves rapidly and can be difficult to detect. [2] Both overdiagnosis and under-diagnosis of ACS carry significant risks.
Overdiagnosis of ACS exposes patients to increased risk of complications. Performing a fasciotomy on a tibia fracture patient will increase their length of stay by 8 days and triple their risk of surgical site infection (SSI) to 25%. [3] SSI is the third most costly type of healthcare-acquired infection (HAI) with an estimated added cost of $23,466 per case. [4]
At the same time, a missed or delayed diagnosis can lead to amputation. [5, 6] Amputations carry a lifetime cost of $600,000 and significant medicolegal liability risk. [7] ACS lawsuit rulings favor the patient 33-55% of the time, and damage awards average more than $1,550,000. [8, 9, 10]
The MY01 Continuous Compartmental Pressure Monitor is the only diagnostic aid that can help physicians avoid both under- and overdiagnosis of ACS.
Brief epidemiology of acute compartment syndrome
Trauma is the most common cause of ACS. Two-thirds of ACS cases follow a fracture, most commonly of the tibial diaphysis. [11] The syndrome develops when injury leads to tissue inflammation and increased pressure within a muscle compartment. Increased intracompartmental pressure leads to reduced blood flow and oxygenation. [12]
The standard treatment for ACS is fasciotomy, which involves making one or more incisions in the muscle fascia to decompress the affected compartments. Early diagnosis is a critical factor in avoiding permanent muscle damage or sequelae, thus ensuring the best patient outcomes. [11, 12]
The risks of undertreatment in ACS are significant. Approximately 12% of ACS cases advance to necrosis in three or more compartments, and approximately 7% of cases are missed. [13] Missed or delayed diagnosis of ACS can lead to severe pain, paralysis, nerve damage, muscle necrosis and amputation. [14] Among patients with combat injuries, incomplete or delayed fasciotomy are both associated with higher rates of muscle necrosis and excision, and delayed fasciotomy is also associated with a higher rate of amputation. [15]
There are also significant risks to overtreatment, and approximately 23% of fasciotomies are unnecessary. [5] Patients who receive unnecessary fasciotomy face an extended recovery period with additional risk of infection and disability. Overtreatment also increases costs due to extended length of stay and an overall increase in average hospital charges. [16]
Diagnosing acute compartment syndrome
Traditionally, diagnosis of ACS is based on clinical assessment using the “6 P’s”: pain, paresthesia, pallor, paralysis, pulselessness and poikilothermia.
However, diagnosis based on clinical signs alone has a low sensitivity (13% to 54%). [5] One challenge is that clinical signs are not all relevant in every stage of ACS. For example, paralysis is a late sign of ACS [17], so its utility for diagnosing this rapidly evolving condition is limited.
The American Association of Orthopedic Surgeons (AAOS) recommends the use of continuous intracompartmental pressure (cICP) measurement in the diagnosis of ACS. [17] Monitoring cICP provides an objective measure that augments subjective clinical assessments. For patients with tibial shaft fracture suspected of ACS, cICP has an estimated sensitivity of 94% and an estimated specificity of 98%. [18]
The MY01 Continuous Compartmental Pressure Monitor uses micro-electro-mechanical system (MEMS) sensing technology to measure cICP. Compared to other compartment pressure measurement devices, the MY01 is significantly more reliable at tracking pressure changes within muscle compartments. [12]
The MY01 is a Class II medical device cleared by U.S. Food and Drug Administration as an aid to diagnosis for ACS. It is also licensed for sale under CE and Health Canada regulations. To use the MY01 device:
1. Identify patient at risk
According to a recent review, [11] the clinical signs of ACS are:
- Swelling
- Pain on passive stretch
- Pain out of proportion to injury
- Paresthesia and sensory changes
- Paralysis and motor changes
According to the same review, risk factors for developing ACS include youth, tibia fracture, high-energy fracture of the femoral shaft or forearm, and polytrauma. [11]
2. Place sensor in muscle compartment
Prep the insertion site. Prepare the patient’s skin as normal using aseptic practices. Consider applying a local anesthetic.
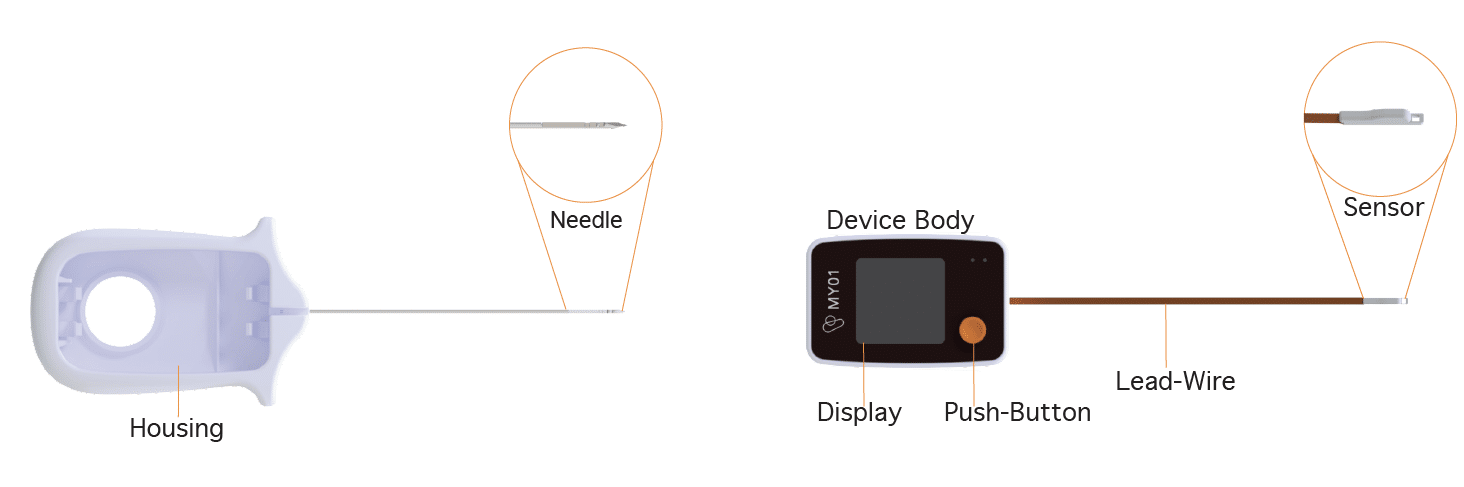
Prep the device. Remove the MY01 device from the packaging. The MY01 consists of two parts — the device body (including the sensor, the lead wire and the display monitor) and the introducer (including the housing and the introducer needle). Press and hold the power button until the display activates.
Insert the sensor. Remove the needle cap. Insert the introducer needle into the muscle compartment in a controlled linear fashion. Use the markings on the introducer to estimate insertion depth. (Note: The MY01 can be used to capture several single-point measurements from up to 5 different insertion sites.)
Remove the introducer. When the display readings have stabilized, eject the display monitor from the introducer by pressing through the opening in the back of the housing. Fix the monitor to the patient’s skin using the exposed adhesive. Gently rotate the introducer needle 180 degrees to disengage the sensor, and then withdraw the needle.
Dress the insertion site. Secure the insertion point with transparent film dressing. (Note: Make sure not to cover the pressure vent on the display monitor.)
3. Monitor patient with combinational approach
The MY01 sensor can stay in the patient for up to 18 hours, and it can remain in place as the patient moves between care units. (Check the device regularly to make sure the sensor remains secure.)
In the diagnosis of ACS, physicians use ICP and the patient’s diastolic blood pressure (DBP) to calculate the perfusion pressure in the affected compartment. [19] The formula is:
DBP – ICP = perfusion pressure
According to an AAOS clinical practice guideline, “a perfusion pressure of greater than 30 mmHg is safe for ruling out compartment syndrome”. [17]
It is recommended to use data from the MY01 sensor in combination with clinical assessment.
Data from the MY01 is archived in a cloud-based server. Physicians can download patient data as needed to support clinical documentation.
In addition, the MY01 Mobile Application is an optional smartphone app intended for storing and displaying identical pressure values from the MY01 monitor device. The data is for informational purposes only and is not intended to be used for diagnosis of any nature or for active patient monitoring
Learn more
The MY01 is manufactured by NXTSENS Microsystems. The company’s vision is to provide physicians with reliable data at their fingertips so they can focus on the patient.
For complete details on how to use the MY01, see full operational instructions. To see the MY01 for yourself, book a trial today.
References
1. Harding, Margaret Meyer MSN, RN, AGPCNP-BC, CPAN Acute compartment syndrome, The Nurse Practitioner: April 2019 – Volume 44 – Issue 4 – p 23-28 doi: 10.1097/01.NPR.0000554087.90202.2b
2. Ulmer, T., The clinical diagnosis of compartment syndrome of the lower leg: are clinical findings predictive of the disorder? J Orthop Trauma, 2002. 16(8): p. 572-7.
3. Dubina AG, Paryavi E, Manson TT, Allmon C, O’Toole RV. Surgical site infection in tibial plateau fractures with ipsilateral compartment syndrome. Injury. 2017 Feb;48(2):495-500. doi: 10.1016/j.injury.2016.10.017. Epub 2016 Oct 18. PMID: 27914662.
4. Zimlichman E, Henderson D, Tamir O, Franz C, Song P, Yamin CK, Keohane C, Denham CR, Bates DW. Health care–associated infections: a meta-analysis of costs and financial impact on the US health care system. JAMA Intern Med. 2013;173(22):2039–46.
5. Lor, K.K.H., et al., Raised compartment pressures are frequently observed with tibial shaft fractures despite the absence of compartment syndrome: A prospective cohort study. J Orthop Surg (Hong Kong), 2017. 25(2): p. 2309499017717362.
6. Cone J, Inaba KLower extremity compartment syndromeTrauma Surgery & Acute Care Open 2017;2:e000094. doi: 10.1136/tsaco-2017-000094
7. Chung, Kevin C et al. “A cost-utility analysis of amputation versus salvage for Gustilo type IIIB and IIIC open tibial fractures.” Plastic and reconstructive surgery vol. 124,6 (2009): 1965-1973. doi:10.1097/PRS.0b013e3181bcf156
8. John Phair, Matthew Carnevale, Larry A. Scher, Karan Garg,
Malpractice Litigation for Compartment Syndrome, Annals of Vascular Surgery, Volume 67, 2020, Pages 143-147, ISSN 0890-5096, https://doi.org/10.1016/j.avsg.2020.04.012 (https://www.sciencedirect.com/science/article/pii/S0890509620303319)
9. Bhattacharyya T, Vrahas MS: The medical-legal aspects of compartment syndrome. J Bone Joint Surg Am 2004;86:864-868.
10. Little M.T.M., Lin C.A., Vrahas M.S. (2019) Legal Aspects of Compartment Syndrome. In: Mauffrey C., Hak D., Martin III M. (eds) Compartment Syndrome. Springer, Cham. https://doi.org/10.1007/978-3-030-22331-1_2
11. Duckworth, A.D. and M.M. McQueen, The Diagnosis of Acute Compartment Syndrome: A Critical Analysis Review. JBJS Rev, 2017. 5(12): p. e1.
12. Merle, G., et al., Comparison of Three Devices to Measure Pressure for Acute Compartment Syndrome. Military Medicine, 2020. 185(Supplement_1): p. 77-81.
13. Boyers, D., Costing report for a new diagnostic test for the detection of acute compartment syndrome., H.E.R. Unit, Editor. 2012, Health Services Research Unit, University of Aberdeen, United Kingdom.
14. Harvey, E.J., et al., What’s new in acute compartment syndrome? J Orthop Trauma, 2012. 26(12): p. 699-702.
15. Ritenour, A.E., et al., Complications after fasciotomy revision and delayed compartment release in combat patients. J Trauma, 2008. 64(2 Suppl): p. S153-61; discussion S161-2.
16. Schmidt, A.H., The impact of compartment syndrome on hospital length of stay and charges among adult patients admitted with a fracture of the tibia. J Orthop Trauma, 2011. 25(6): p. 355-7.
17. Osborn, C.P.M. and A.H. Schmidt, Management of Acute Compartment Syndrome. J Am Acad Orthop Surg, 2019.
18. McQueen, M.M., et al., The estimated sensitivity and specificity of compartment pressure monitoring for acute compartment syndrome. J Bone Joint Surg Am, 2013. 95(8): p. 673-7.
19. Schmidt, A.H., Acute Compartment Syndrome. Orthopedic Clinics of North America, 2016. 47(3): p. 517-525.